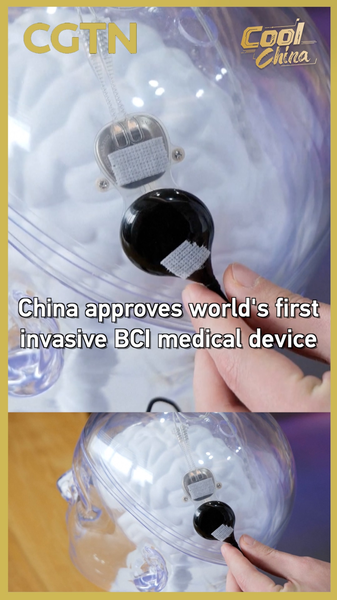
China has made medical history by greenlighting the world's first invasive Brain-Computer Interface (BCI) device for clinical applications. The National Medical Products Administration (NMPA) recently approved Neuracle Medical Technology's implantable system designed to restore hand motor functions, signaling a new era in neurorehabilitation technology.
The Shanghai-developed device translates neural signals into digital commands through implanted electrodes, offering hope for patients with paralysis caused by spinal cord injuries or stroke. Clinical trials demonstrated significant improvement in patients' ability to perform daily tasks through direct brain signal interpretation.
"This approval represents a quantum leap in merging human neurology with digital systems," said Dr. Li Wei, a lead researcher at Tsinghua University's Neuroengineering Center. Medical professionals anticipate the technology could eventually address conditions ranging from Parkinson's disease to chronic pain management.
For investors, this breakthrough highlights China's growing dominance in advanced medical technologies. The BCI market in Asia is projected to exceed $2 billion by 2028, with applications expanding into rehabilitation medicine and cognitive enhancement.
While ethical discussions continue about neural interface technologies, the NMPA's rigorous approval process ensures compliance with international medical safety standards. This milestone positions the Chinese mainland as a pioneer in next-generation neurotechnological solutions.
Reference(s):
cgtn.com