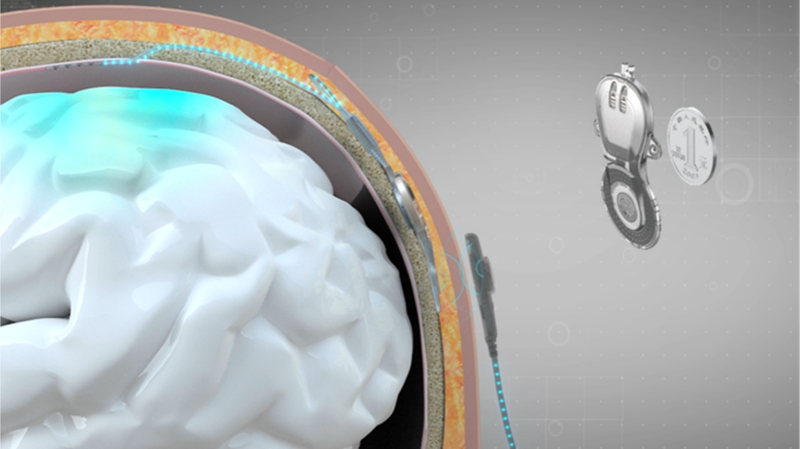
China has achieved a landmark breakthrough in neurotechnology with the commercial approval of the world's first invasive brain-computer interface (BCI) medical device this month. The National Medical Products Administration greenlit the implantable system on March 13, 2026, developed through collaboration between Shanghai's Neuracle Medical Technology and Tsinghua University.
This revolutionary technology decodes neural signals from motor-related brain regions through implanted electrodes, translating thoughts into actionable commands. Designed specifically for patients with cervical spinal cord injuries causing tetraplegia, the system enables basic hand functions through a neural-controlled pneumatic glove – restoring simple but life-changing abilities like grasping objects.
While non-invasive BCIs have seen global development, China's successful commercialization of an implantable system positions it at the forefront of medical neurotech. The approval follows extensive clinical trials demonstrating improved quality of life for participants, with 89% showing measurable improvement in daily task completion during final testing phases.
Industry analysts project the global BCI market could reach $3.8 billion by 2028, with medical applications driving 62% of growth. This development comes as China implements its 15th Five-Year Plan emphasizing healthcare technology innovation, signaling potential for expanded applications in neurological rehabilitation and assistive technologies.
Reference(s):
cgtn.com