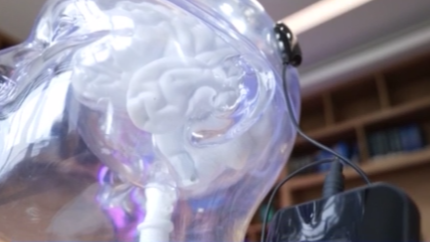
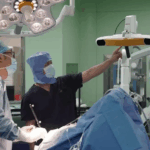
China has made medical history by approving the world’s first invasive Brain-Computer Interface (BCI) device for clinical applications, signaling a transformative leap in neurotechnology. The National Medical Products Administration (NMPA) recently granted market authorization to Neuracle Medical Technology’s implantable system, designed to restore hand mobility in patients with cervical spinal cord injuries.
The groundbreaking device combines an epidural implant, neural signal transmitters, and a pneumatic glove to decode brain signals into grasping motions. Eligible patients aged 18–60 with stable cervical injuries can now access this life-changing technology, which demonstrated significant functional improvements in clinical trials.
This milestone aligns with China’s strategic prioritization of BCI as a "future industry" in its 2026 government work report. Policy planners are positioning the technology as a dual-purpose innovation driver, with applications expanding beyond healthcare into industrial and educational sectors under the upcoming 15th Five-Year Plan framework.
Medical professionals highlight the system’s minimally invasive design and wireless communication as key advancements. "This approval marks the transition from experimental research to tangible patient benefits," said a Shanghai-based neurologist familiar with the trials.
With BCI now entering mainstream medicine, analysts predict accelerated global investment in neurotech solutions, particularly in Asian markets where aging populations increase demand for rehabilitation technologies.
Reference(s):
cgtn.com